The fund that finds them first
BVF Partners is a San Francisco-based private investment partnership specialising in fundamentally-driven public biotechnology investments. Since its inception in 1993, it has strived to build concentrated, long-term positions in small-cap biotechnology companies while performing rigorous diligence and ongoing monitoring.
That description understates what BVF actually is. What BVF is, is a thirty-year track record of finding the overlooked company with the genuinely differentiated science - the one the market hasn't priced properly yet - and sitting in it with conviction until the world catches up.
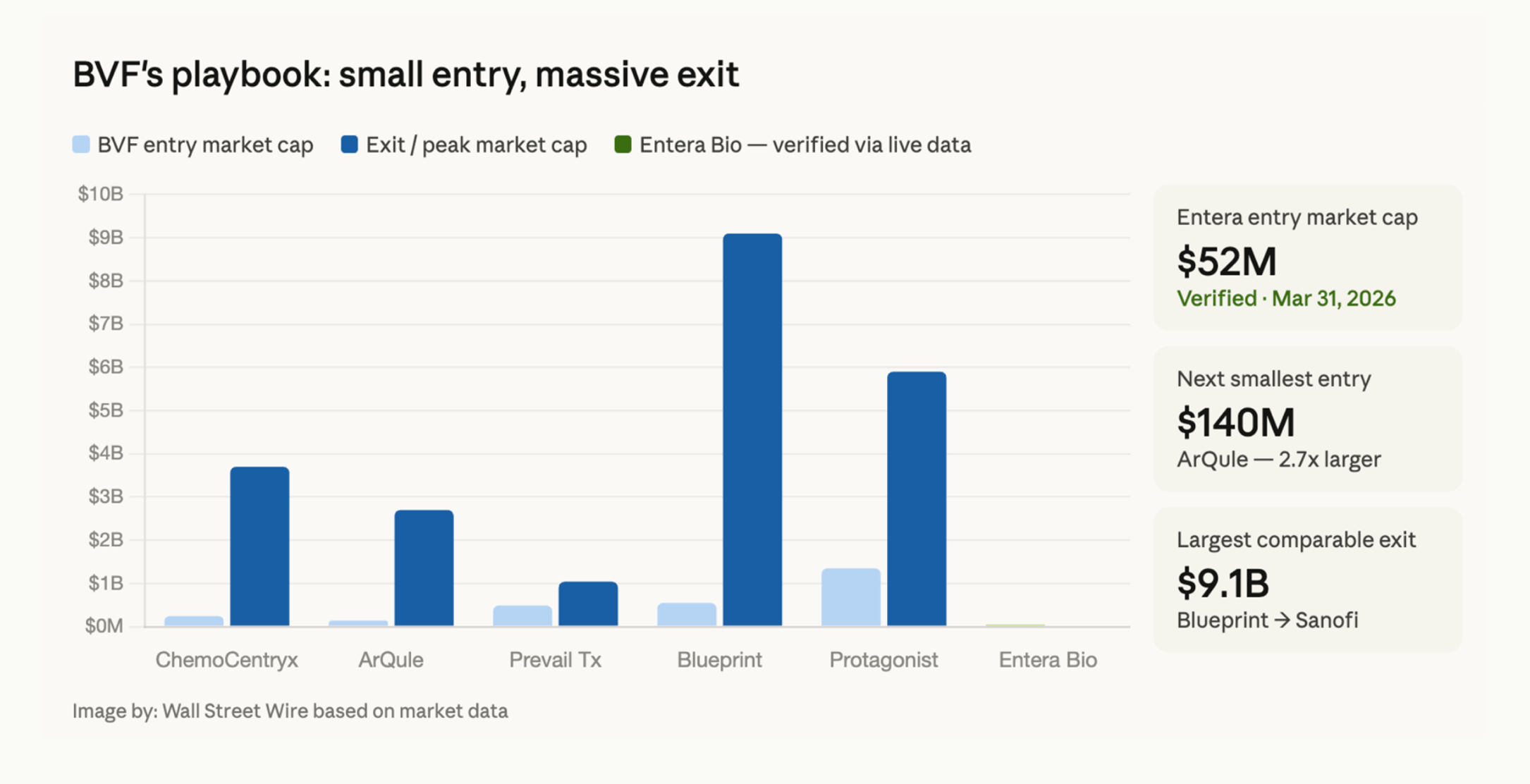
The list reads like a greatest hits of the last decade in pharma M&A. ChemoCentryx: acquired by Amgen for $3.7 billion at a 115% premium. Blueprint Medicines: acquired by Sanofi for $9.5 billion. Protagonist Therapeutics: a peptide platform company in which BVF built a $69 million position - its single largest portfolio weighting - that subsequently attracted J&J acquisition talks at over $4 billion. ArQule: Merck paid $2.7 billion at a 100% premium. Kymera: BVF led the Series C before the IPO, and a $150 million Sanofi upfront and $2 billion-plus in potential milestones followed.
The pattern, repeated: BVF invests when the market hasn't caught up with what the science actually says. The market eventually does. The premiums are enormous.
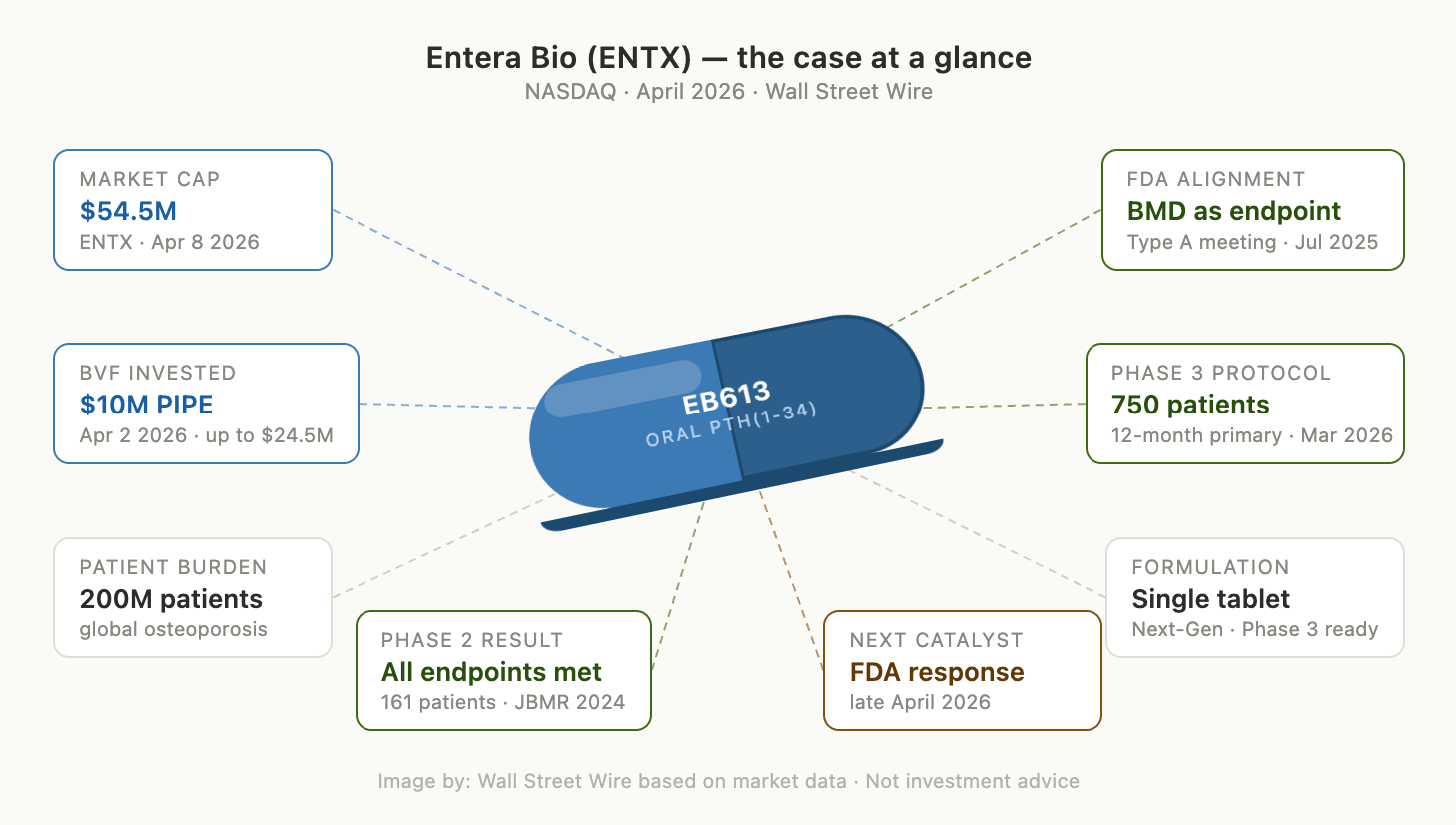
Now BVF has invested in Entera Bio at a market cap of roughly $60 million. BVF led a $10 million private placement structured with five-year warrants to purchase additional shares at $1.24 - with total potential proceeds of up to $24.5 million. Those warrants are not decoration. BVF bought the right to own significantly more at roughly today's price, for five years. That is a statement about what they believe this story has ahead of it.
What they are actually betting on
More than 200 million women worldwide live with osteoporosis. The most effective treatments - anabolic therapies that rebuild bone - have existed for years. Eli Lilly's Forteo generated nearly $1.7 billion in peak annual sales. The data is overwhelming. And yet the majority of patients who would benefit have never received anabolic therapy. The reason is devastatingly simple: injection. Daily subcutaneous injections produce adherence rates below 10%. Doctors are reluctant. Patients delay until the fracture has already happened.
The treatment gap in osteoporosis is not a scientific failure. It is a delivery failure.
Entera's EB613 is designed to close it: an oral once-daily tablet formulation of PTH(1-34) - the same bone-building hormone as Forteo - delivered via the company's proprietary N-Tab® platform. A 161-patient Phase 2 study met its primary pharmacodynamic endpoints and secondary bone mineral density endpoints, producing rapid dose-proportional increases in markers of bone formation, reductions in bone resorption, and significant BMD gains at the lumbar spine, total hip, and femoral neck. The Phase 2 data, published in the Journal of Bone and Mineral Research, showed results comparable to published injectable teriparatide data at the same six-month timepoint.
The mechanism works. Phase 3 will determine whether it crosses the regulatory finish line.
The regulatory jailbreak
For close to a decade, osteoporosis drug development was frozen — not for lack of molecules, but because traditional Phase 3 trials required fracture incidence as the primary endpoint, meaning 2,500 to 14,000 patients, followed for years, at enormous cost. No new osteoporosis drug has been approved since 2019.
Entera spent years making the public scientific case for a better framework. In July 2025, the FDA provided written alignment — in a Type A meeting response — that a single BMD-based Phase 3 trial could support an NDA for EB613. In December 2025, the FDA extended this framework across the entire field, formally qualifying total hip BMD as a validated surrogate endpoint for all novel osteoporosis drugs.
In March 2026, the company submitted a streamlined Phase 3 protocol to the FDA: a 750-patient, multinational, randomised, double-blind, placebo-controlled study evaluating total hip BMD change at 12 months as the primary endpoint, with a 12-month open-label extension running in parallel. Crucially, the drug going into Phase 3 is the final commercial formulation - the single Next-Gen tablet, already bridged to the earlier multi-tablet candidate and to Forteo itself. The asset being tested is the asset that would reach the market. FDA feedback on this evolved protocol design is expected in the coming weeks. The response will be consequential, and BVF is betting it will be positive.
The timing is everything
In each of BVF's most memorable investments, they entered at the last moment before the value inflection made entry prohibitively expensive. With ChemoCentryx, they held through a trough below $2 a share. With ArQule, through a brutal Phase 1 data setback that sent the stock under $3. With Protagonist, they built their largest single weighting just as Phase 2b had confirmed the oral peptide mechanism and Phase 3 was about to begin.
Entera sits at approximately a $60 million dollar market cap. It has completed a Phase 2 with all endpoints met. It has regulatory endpoint alignment and is now pursuing a streamlined trial design. It has the commercial formulation ready. It has a chairman who previously ran Pfizer's $14 billion Global Innovative Pharmaceutical Business. It has a CEO and board buying their own stock in the open market. And it has BVF - the fund that found all of the above before the world noticed.
The bones of the bet are clear. The science seems to work. The path is defined. The market may not have done the arithmetic yet. BVF has seen this pattern before. They know how it tends to end.
Subscribe to Get our Exclusive Insights to your Inbox
=
Recent News Highlights from Entera
=
Important Disclaimers and Disclosures: The author, Wall Street Wire, which CapNotes is an an affiliate of, is a content and media technology platform that connects the market with under-the-radar companies. The platform operates a network of industry-focused media channels spanning finance, biopharma, cyber, AI, and additional sectors, delivering insights on both broader market developments and emerging or overlooked companies. Wall Street Wire is not a broker-dealer or investment adviser. References to market size estimates, valuations, price targets, or other third-party data are provided strictly for informational purposes. Wall Street Wire receives cash compensation from Entera Bio Ltd Ltd for coverage and awareness services, which are provided on an ongoing subscription basis. The content above is a form of paid advertising and promotion and is for informational purposes only and does not constitute financial or investment advice. Full compensation details, information about the operator of Wall Street Wire, and the complete set of disclaimers and disclosures applicable to this content are available at: wallstwire.ai/disclosures. This article should not be considered an official communication of the issuer.